Leveraging the unique biological advantages of zebrafish, this platform utilizes chemical (testing of small molecule compounds), physical (mechanical injury), biological (microbial infection and cell transplantation), and molecular biology (transgenic and gene editing) techniques to rapidly engineer precise models for various diseases including cancer, neurodegeneration, inflammation, and musculoskeletal disorders.
Advantages of zebrafish as a disease model:
· Multidimensional modeling: ability to create models through chemical induction and genetic modification that support a complete research path from phenotype simulation to mechanistic studies.
· High throughput screening: Small and large offspring numbers, zebrafish can be used for high-throughput screening in 96 well plates. Drug efficacy and toxicity screens are quick and convenient.
· Accurate efficacy evaluation: Transparent embryo characteristics, multiple mutantant strains, combined with in vivo imaging technology, zebrafish can be tracked for drug efficacy and synchronous observation of multiple developing organs.
1. Tumor disease models
The in vivozebrafish tumor transplantation model has become an important tool for cancer research. Due to its ease of operation, low cost, and high throughput it is especially suitable for preclinical evaluation and early screening of anti-caner drugs. Fluorescent labeled human tumor cells can be implanted into transparent zebrafish embryos via microinjection and followed with in vivo imaging to allow rapid and high-throughput dynamic monitoring of tumor proliferation and metastasis.
Types of Zebrafish xenograft models:
· Cell line-derived xenograft,CDX
· Patient derived xenograft,PDX
[Service Contents ]
zCDX;zPDX;Tumor microenvironment analysis; Anti-cancer therapy; Quick and personalized assessment of anti-cancer drug efficacy in Precision Medicine.
[Case]
[Case]
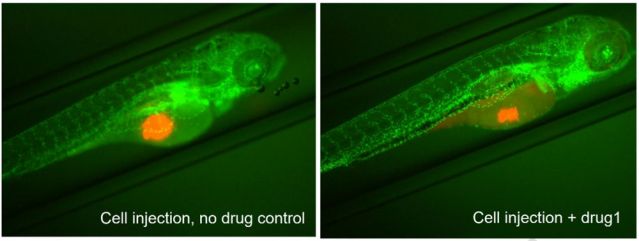
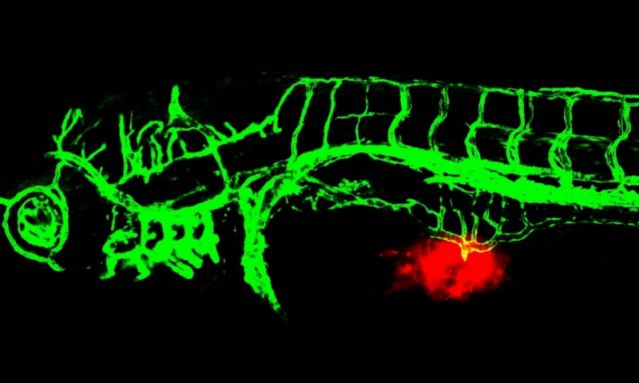
Red fluorescently labeled tumor cells are injected into the Tg (fli1: EGFP) transgenic zebrafish embryo models to observe their effects on angiogenesis in real-time. This model can visualize the interaction between tumor microenvironment and angiogenesis, providing an efficient and accurate in vivo method for screening anti-tumor drugs and studying drug mechanisms.
2. Cardiovascular and cerebrovascular disease models:
Genetically modified zebrafish strains such as Tg (fli1a: GFP) and Tg (flk1: EGFP) allow visualization of their entire vasculature. Changes in angiogenesis can be visualized via fluorescence microscope, which is simple and fast. The molecular mechanism of zebrafish vascular development is highly similar to that of mammals, making it very suitable for evaluating the efficacy of anti-angiogenic drugs and using such zebrafish models for drug screening.
[Service Areas]
Heart failure models; Thrombosis formation; Angiogenesis; Drug efficacy and toxicity screens.
[Case]
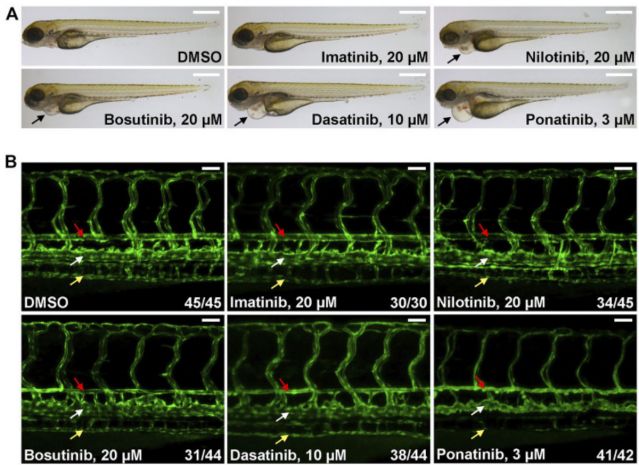
SSome tyrosine kinase inhibitors used to treat chronic myeloid leukemia induce cardiovascular toxicity.
[Case]
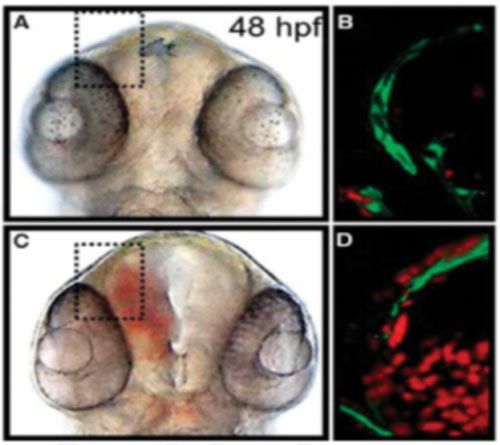
Zebrafish model of hemorrhagic stroke.
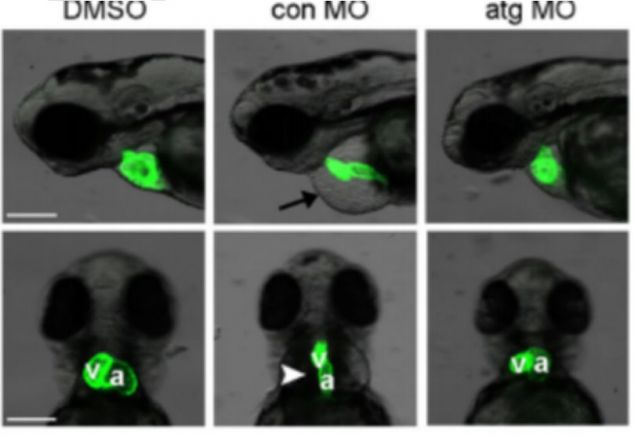
Zebrafish model of heart failure
3. Disease models of blood system:
Zebrafish are an ideal model for studying drugs targeting blood disorders since the zebrafish blood system is very similar to that of humans with highly conserved coagulation factors and platelet receptors . Blood circulation of zebrafish fry can be directly observed under a stereomicroscope using transgenic zebrafish strains with fluorescently labeled blood cells or using dyes that stain blood cells, allowing rapid evaluation of drug efficacy and drug screening for blood disorders.
[Service Areas]
Anemia model (induced by phenylhydrazine compounds); Leukemia model; Thrombocytopenia (induced by antiplatelet serum); Genetic model of thrombocytopenia: stx12 KO
[Case]
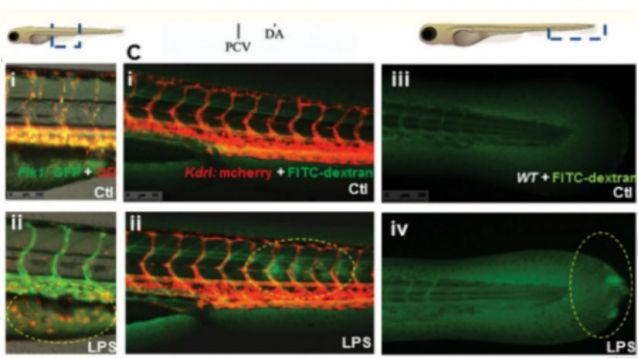
Severe sepsis model
4. Neurological Disease Models:
With its high genetic homology to the human nervous system and a rapid developmental cycle, the zebrafish is an ideal model for assessing neuropharmaceutical efficacy and studying neurodegenerative diseases. Transgenic zebrafish strains with fluorescently labeled motor neurons/glial cells already exist.
[Service Areas]
Parkinson's disease (MPTP/6-OHDA induction); ALS (transgenic zebrafish expressing FUS, TDP43 gene mutations); Epilepsy (scn1lab mutation).
[Case]
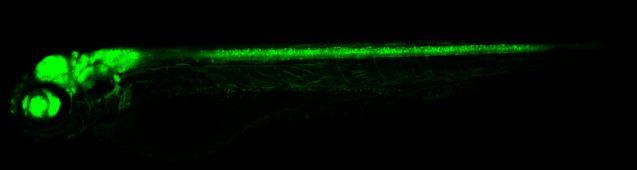
Tg (HuC: EGFP) transgenic zebrafish drives EGFP fluorescent protein expression through HuC neuron specific promoters, which can visually label mature neurons during development. It is suitable for dynamic observation of neural development, damage repair, and neurotoxicity in vivo, and is an efficient visualization model for neuroscience research and drug screening.
5. Liver and kidney disease models:
Zebrafish liver shares homologous lipid metabolism enzymes and detoxification mechanisms with mammals, making it suitable for drug research. Available liver fluorescence strains (such as Tg (lfabp: EGFP)) enable tracking of drug-induced changes in fluorescence of liver tissue through in vivo imaging. The efficacy indicators include: pathological grading of hepatic steatosis/fibrosis, delayed absorption of yolk sac (related to liver function), and organ section analysis (Oil Red O/Sirius Red staining). These models can be efficiently used to screen for hepatoprotective drugs and evaluate liver toxicity.
[Service Areas]
Fatty liver (ethanol induced); Cirrhosis (CCl4 induced; TGF-β1 transgenic); Glomerular injury (induced by gentamicin); Hepatorenal Syndrome (Hereditary Polycystic Kidney Model)
[Case]
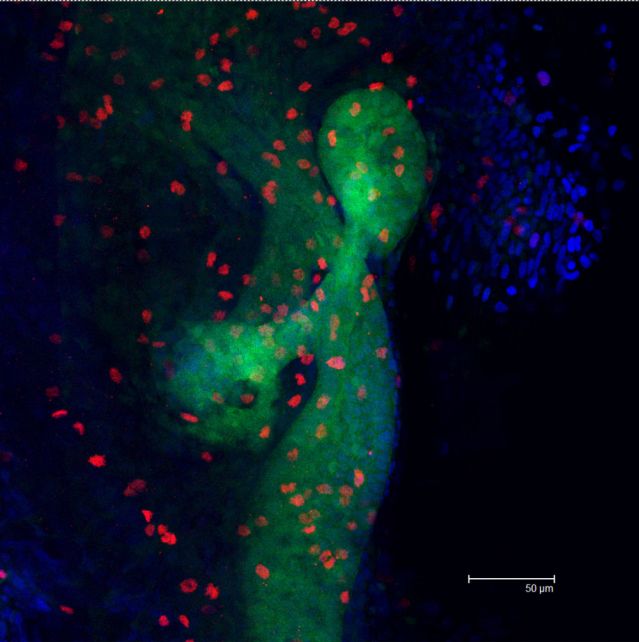
In the transgenic zebrafish strain Tg(gut:GFP), green fluorescence is used to label intestinal epithelial cells (GFP), red fluorescence shows the cell proliferation area labeled by immunostaining of phosphorylated histone H3(pH3) antibody, and blue is DAPI nuclear staining.
6. Skeletal and muscular disease models:
The zebrafish skeletal and muscular models can be established quickly and support high-throughput drug screening. Based on the glucocorticoid induced osteoporosis model, which is highly similar to human pathology, rapid in vivo efficacy evaluation can be carried out in microplates. By using specific fluorescent labeling techniques such as osteocalcin/Alzarin Red staining, the changes in skull bone density can be accurately measured, providing a sensitive quantitative analysis platform for anti-osteoporosis drugs.
[Service Content]
Osteoporosis (dexamethasone induced); Muscle atrophy (induced by prednisolone); Osteoarthritis (induced by sodium iodoacetate); Osteogenesis imperfecta (col1a1 gene mutation)
[Case]
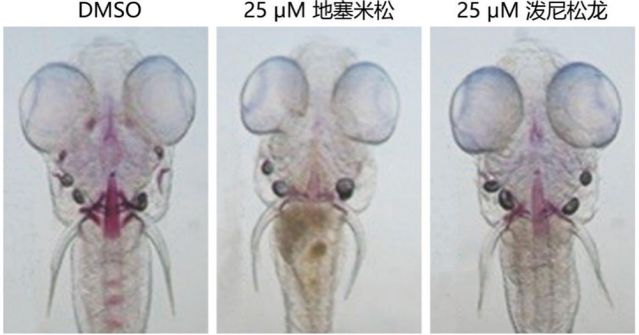
A zebrafish osteoporosis model induced by dexamethasone and prednisolone glucocorticoids with specifically labeled calcified hard bone structures (red) using Alizarin Red S staining.
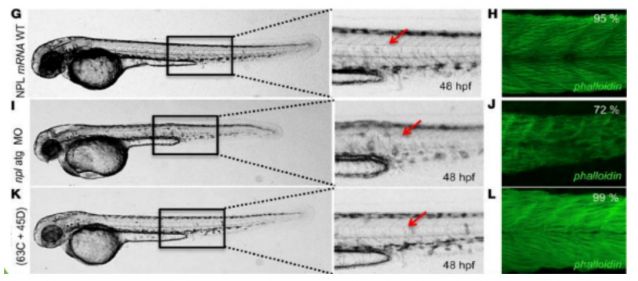
Muscle degenerative disease model
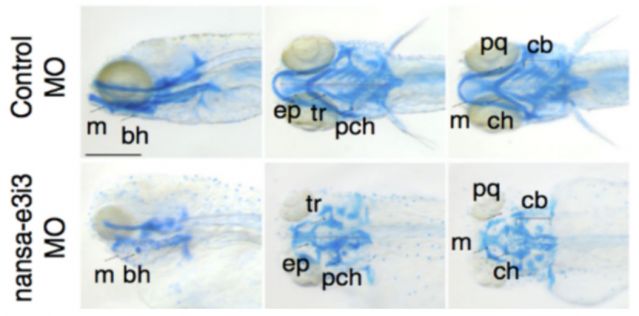
Skeletal Dysplasia
7. Inflammatory and immune disease models:
Zebrafish larvae are transparent and have a highly conserved immune system compared to humans, making them an ideal model for studying inflammation and immune diseases. Labeling of neutrophils and macrophages in transgenic strains such as Tg(mpx:EGFP) and Tg (mpeg1: EGFP) allow real-time tracking of immune cell migration and inflammatory response. The expression of inflammatory factors (TNF-α/IL-1b), tissue pathology, and drug intervention effects can be quantitatively analyzed through physical (tail fin cutting)/chemical (lipopolysaccharide) or gene editing (il1 β/NF - κ B mutation) models.
[Service Areas]
Acute inflammation (lipopolysaccharide LPS induction; caudal fin cutting); Septicemia model (induced by bacterial infection); Immunodeficiency (rag1 gene knockout); Arthritis (induced by carrageenan);
[Case]
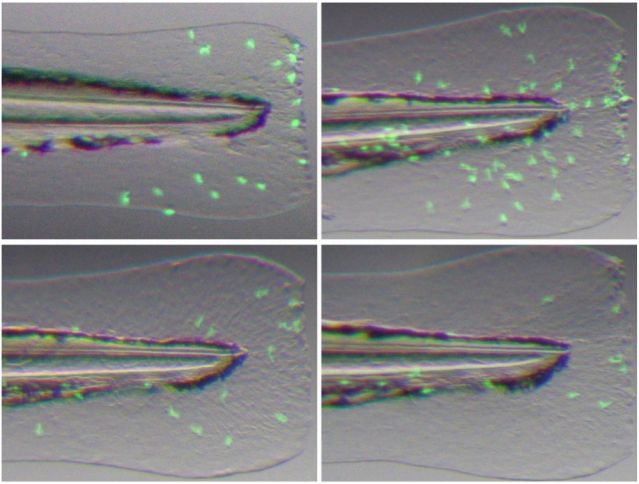
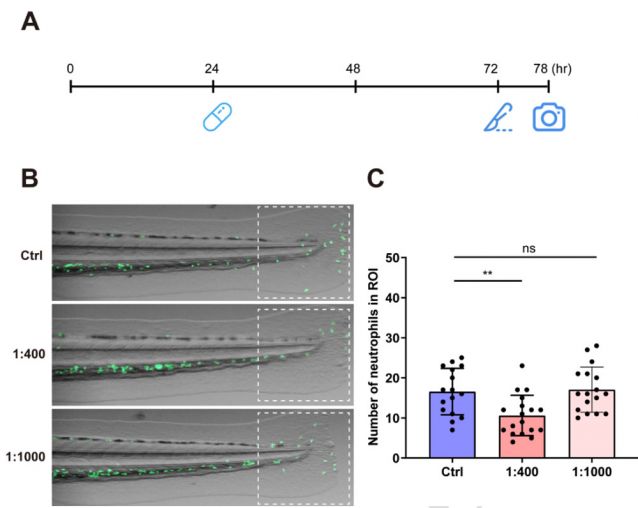
Based on the Tg (mpx:EGFP) transgenic zebrafish model (green fluorescent labeled neutrophils), an inflammatory model caused by physical damage was established by precise surgical removal of the caudal fin. Microscopic imaging and dynamically tracking of neutrophils showed their chemotactic migration, infiltration, and the inflammation resolution process at the site of injury.
[Case]
8. Auditory Disease Models:
The auditory system of zebrafish consists of two main sensory structures, the inner ear and the lateral line. Zebrafish have a typical inner ear structure, and the hair cells in the inner ear and lateral line are very similar in structure and function to those in mammals, making it suitable for the study and efficacy testing of auditory cell drugs.
[Service Areas]
Hair cell injury (induced by neomycin); Ototoxic injury (cisplatin induced); Usher syndrome (ush2a gene mutation);
[Case]
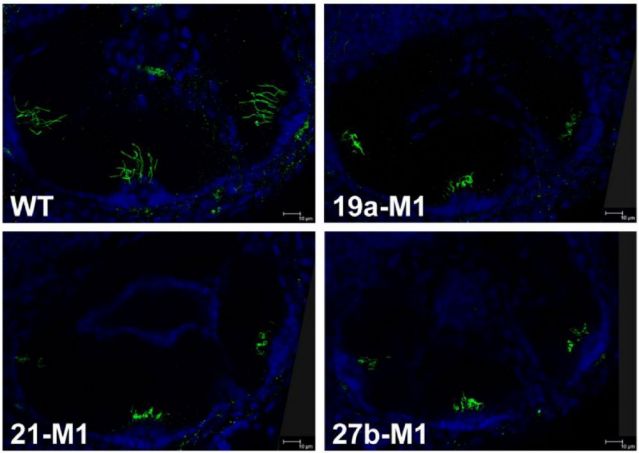
The cilia structure of zebrafish inner ear hair cells was imaged by double labeling with anti-acetylated tubulin immunofluorescence staining (green, labeled with cilia axon microtubules) and DAPI nuclear staining (blue). Based on confocal Z-stack 3D reconstruction technology, cilia length/density can be accurately quantified, which is suitable for evaluating ototoxic drug damage, studying cilia regeneration mechanisms, and constructing genetic hearing loss models.
9. Ophthalmic disease models:
The eye structure of zebrafish is highly conserved compared to humans, including regulation of lens transparency, retinal stratification (photoreceptors/bipolar cells), angiogenesis (such as the retinal vascular network), and optic nerve connections. Zebrafish embryo transparency allows for in vivo observation of eye development or pathological processes (such as cataracts, glaucoma) under drug intervention. By chemical induction (such as monosodium glutamate induced retinal damage) or gene editing (crx/rho mutation mimicking hereditary eye disease), combined with vascular/retinal fluorescent strains such as Tg (flk1: EGFP), the effects of drugs on vascular proliferation, optic nerve protection, or photoreceptor regeneration can be quantitatively evaluated. Such models are suitable for efficient screening drugs for anti-VEGF effects, neurotrophic factors, and as antioxidants.
[Service Areas]
Cataract (induced by sodium selenite); Diabetes retinopathy (induced by high glucose); Hereditary retinitis pigmentosa (rho gene knockout); Retinal vascular hyperplasia (VEGF overexpression).