ZebraPeutics provides gene editing services to create zebrafish disease models: Using CRISPR/Cas9 technology, endogenous genes can be efficiently and quickly knocked out in zebrafish to study gene function and establish disease models. This approach is highly suitable for drug discovery and mechanistic studies in fields such as neurodegenerative, cardiovascular, metabolic diseases. Relying on efficient and precise base editing technology, precise targeted mutations can be achieved at the single nucleotide level, providing innovative solutions for modeling human genetic diseases and fine regulation of gene function. At the same time, customized transgenic model construction is provided, and fluorescent labeling technology is used to achieve visual tracking of live tissues, supporting dynamic biological observation from embryonic to juvenile stages.
The Zebrafish Gene Editing Platform features:
· Rapid R&D: Complete phenotype screening within 1-2 week after gene knockdown, and construction of stable strains within 6 months.
· Large quantity model preparation: The platform can deliver batches of gene editing models, meeting the needs of large-scale experiments.
· Visual analysis: Combining the embryonic transparency with in vivo imaging technology, it achieves dynamic tracking of live cells from embryos to juvenile stages, supporting dynamic monitoring of gene expression.
1. Gene editing service process:
· Project design
· sgRNA testing
· Donor design
· Micro-injection
· Clutch verification
· Germline verification
2. Gene editing services:
(1)Knockout
Gene knockout via CRISPR/Cas9 technology involves the design of sgRNA targeting the target gene sequence to guide the Cas9 nuclease to induce double strand DNA breaks at specific sites. During the process of repairing broken DNA, base mutations or insertions/deletions (indels) of the target gene are introduced into the zebrafish genome, ultimately leading to functional loss of the target gene and achieving the goal of gene knockout, which can effectively delete large fragments of genomic DNA.
[Case]
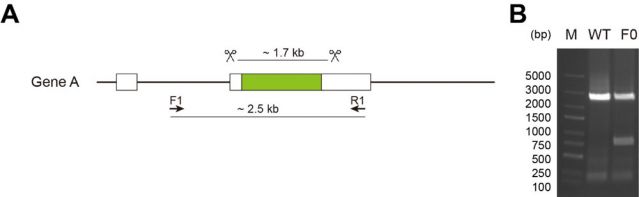
It is necessary to accurately design dual target sgRNA targeting gene coding regions (CDS) at both ends, optimize editing efficiency through efficient screening, and achieve full reading frame knockout of target gene to ensure complete functional inactivation.
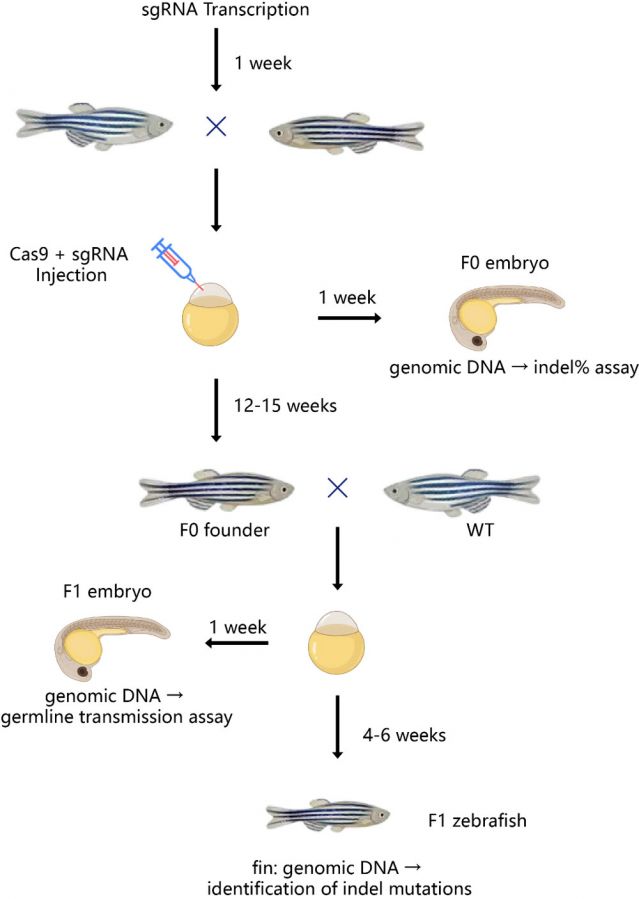
Schematic diagram of the zebrafish gene knockout service process
(2)Transgene
Using the Tol2 transposon system, a vector containing specific promoters to drive the expression of target genes is constructed and injected together with transposase mRNA into zebrafish single-cell embryos to construct transgenic zebrafish. This allows construction of transgenic zebrafish with inducible and tissue-specific expression of target genes using the TetOn system.
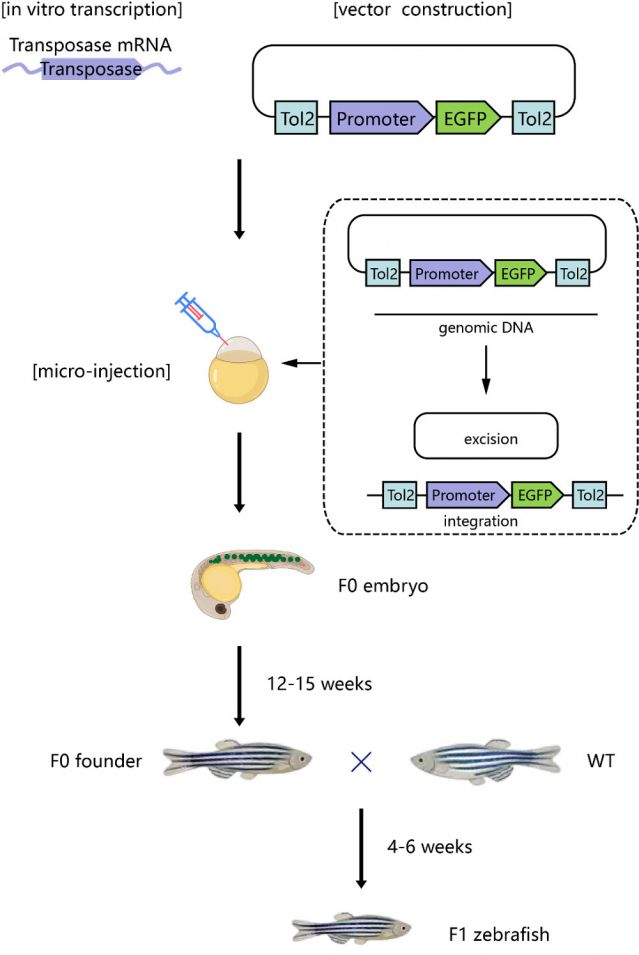
Schematic diagram of zebrafish transgenic service process
(3)Point Mutation
CRISPR/Cas9 technology allows site directed mutagenesis of zebrafish strains. Site directed mutagenesis can be either mediated by base editors ABE/KBE or by homologous recombination donors. Point mutation service is used for modeling genetic diseases caused by point mutations or studying the function of SNPs.
[case]
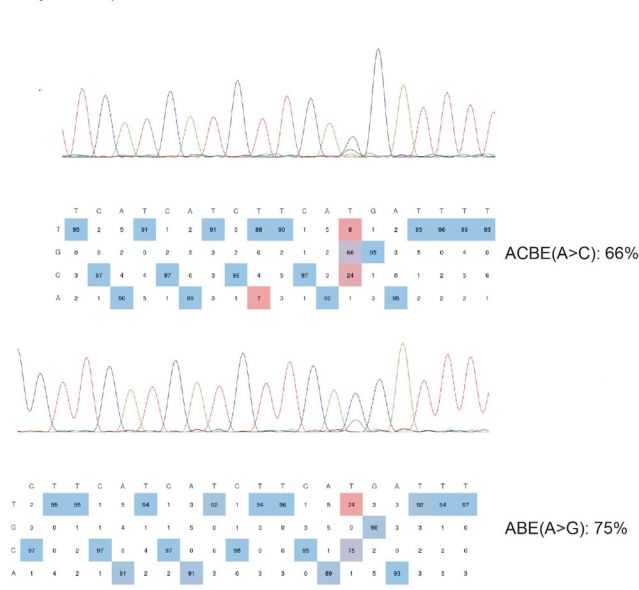
(4)Knockin
Gene knockin services utilize CRISPR/Cas9 technology to construct zebrafish strains with target gene Knockins. Exogenous gene fragments are inserted at specific locations for precise labeling of endogenous gene expression patterns or to achieve temporal and spatial control of gene expression.